Experience, Success and the Best All-Inclusive Price in the Southeast.
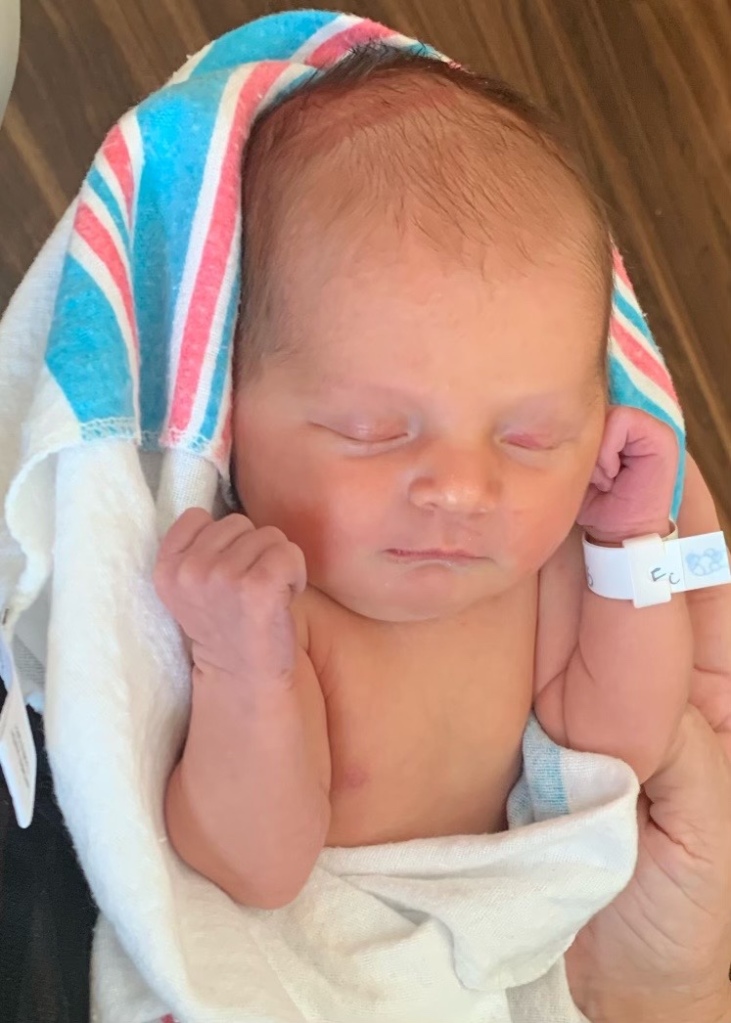
GA Vasectomy Reversal Success | Dr. McHugh is one of the Southeast’s most experienced microscopic vasectomy reversal urologists. Hundreds of successful reversals have been performed in our accredited and dedicated vasectomy reversal center.
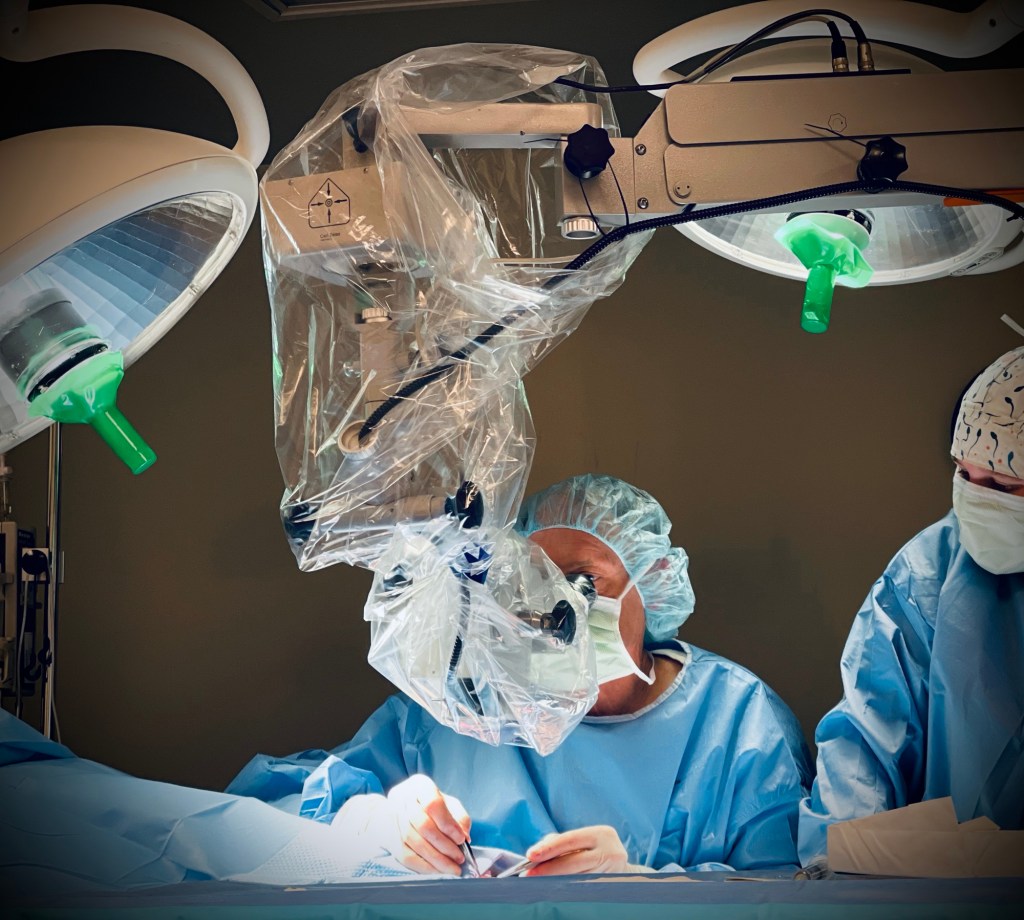
Vasectomy reversals are performed on a weekly basis in our practice owned and on-site surgery center with general anesthesia administered by certified anesthesiologists.
Dr. McHugh’s eBook on Reversals: Everything you’d want to know.

“It worked. Thank you!”
B. and B. H.
“Congratulations! We are very happy for you and your family. Thank you for allowing us to participate in this blessing.”
John McHugh

Click to see some of Dr. McHugh’s success stories!

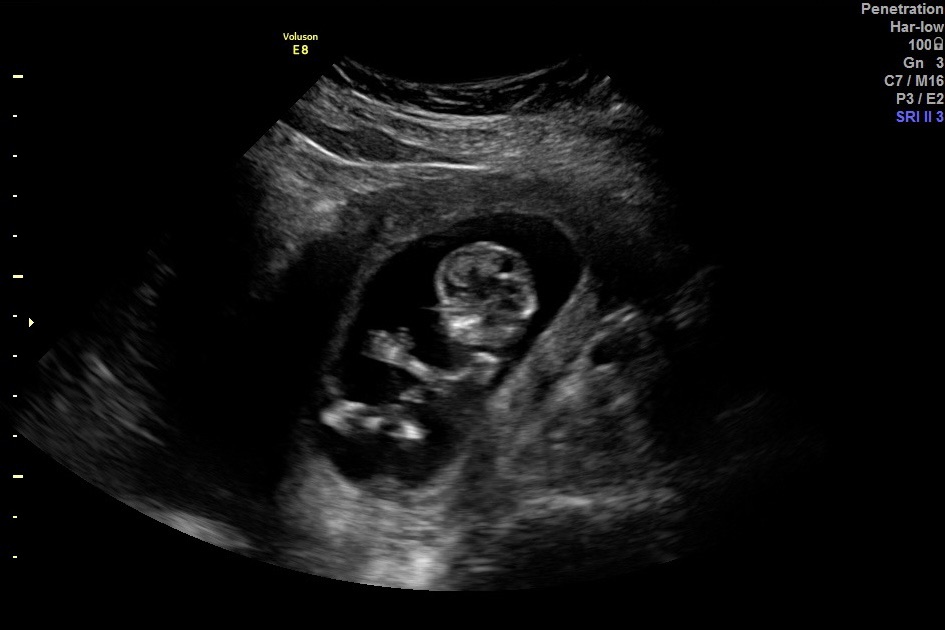
Considering a Vasectomy Reversal? We’d love to help.
Ready to book a reversal consultation? Use our vasectomy reversal online scheduler: You book and Dr. McHugh calls you!
Reversals are what we do!

Did you know?
According to European folklore, the white stork is responsible for bringing babies to new parents. The legend is very ancient, but was popularised by a 19th-century Hans Christian Andersen story called “The Storks.” German folklore held that storks found babies in caves or marshes and brought them to households in a basket on their backs or held in their beaks. These caves contained adebarsteine or “stork stones”.